Updated 26 November 2025
In a previous post I talked about the four forces which govern the way the Universe works. I’m now going to give some examples of fine tuning. How different our Universe would be if these four forces had very different relative strengths.
Summary of the Four Forces
The Universe is governed by four forces (sometimes known as the four interactions).

What would happen if gravity were much stronger or weaker?
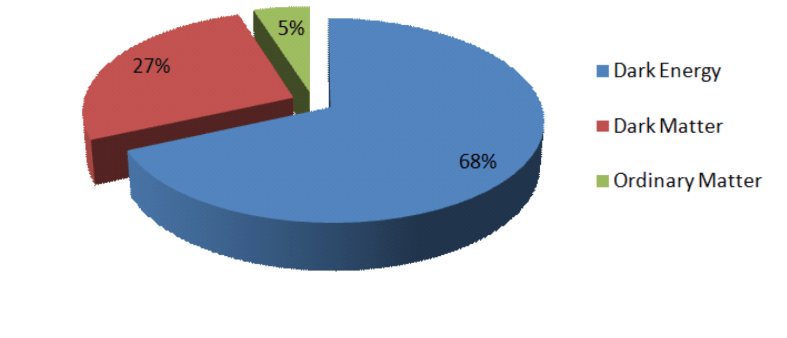
If we average out over the entire observable Universe, then its average mass density is 9 x 10 -27 kg/m3 – an incredibly low value due to the fact that the Universe is mostly empty space. It is made up as follows
Gravity is 1039 times weaker than the electrostatic force and is so weak that it is totally insignificant when dealing with everyday objects having a mass of the order of one kilogram. It plays no part in nuclear physics (which is governed by the other three forces). Chemical bonding and the forces between molecules are governed by the electrostatic force only. The strength of gravity is determined by the gravitational constant G (usually called “big G”), which has the value 6.674 x 10 -11 in standard units.
In a hypothetical universe where gravity were one thousand times stronger (i.e. G 1000 times larger), nuclear physics would be exactly the same as in our Universe. Chemistry and bonding between molecules wouldn’t change either. However, the physics of large objects which are held together by gravity would be very different. Stars would need 30000 times less mass to become hot and dense enough for nuclear reactions to start. (This is because the amount of mass needed to form a star scales as G to the power of -1.5). These mini stars would have much shorter lifetimes, typically a few million years, rather than the 10-billion-year lifetime which our Sun has.
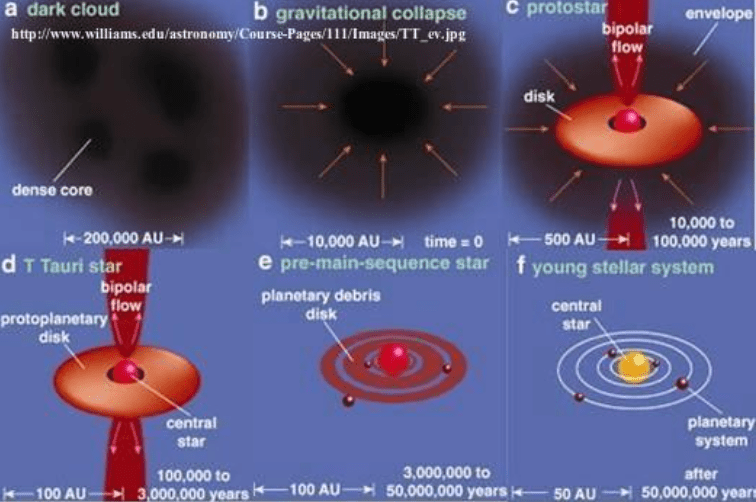
If we were to go even further, and make G one million times larger, the lifetime of stars would be even less- typically thousands of years. In such a universe there just wouldn’t be enough time for the evolution of life to have occurred .
Conversely, if gravity were significantly weaker compared to the other forces, the whole process of the collapse of matter into a dense molecular cloud, which continues to contract until it becomes hot and dense enough for nuclear reactions to start, would take much longer. The amount of time needed to form any star in this ‘low gravity universe’ would be longer than the age we observe for our Universe (13.8 billion years). When they eventually formed such stars would be more massive and burn more slowly and have lifetimes in trillions of years.
If gravity were weaker still, no collapse into stars would occur. Without anything to illuminate it, such a universe would be completely dark.
The Strength of the electrostatic force vs the nuclear force
Atomic nuclei are held together by the residual strong interaction which is more commonly known as the nuclear force. This binds together nucleons (i.e. protons and neutrons) and is very short range. The nuclear force does not follow an inverse square law and falls rapidly with distance – effectively dropping to zero at 3 x 10 -15 metres. It becomes repulsive at distances less than 8 x 10 -16 metres.
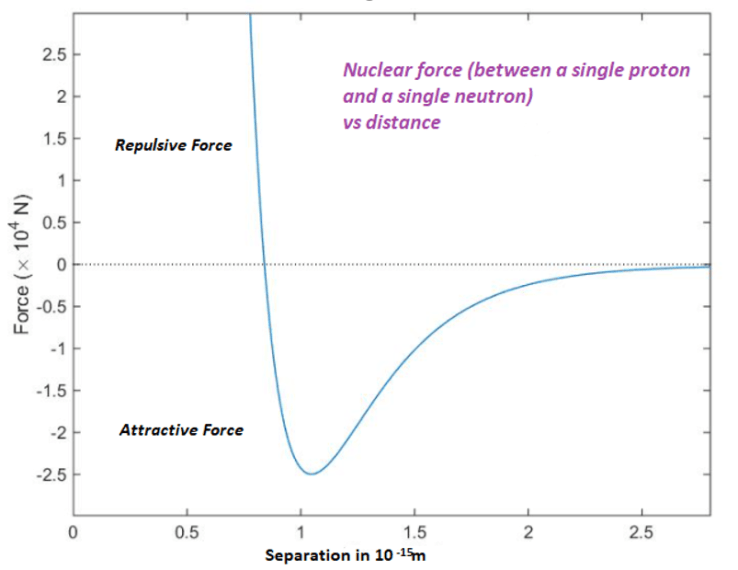
The other important force in the atomic nucleus, the electrostatic force, causes positively charged protons to repel each other. Neutrons, which have zero electric charge, are unaffected by the electrostatic force.
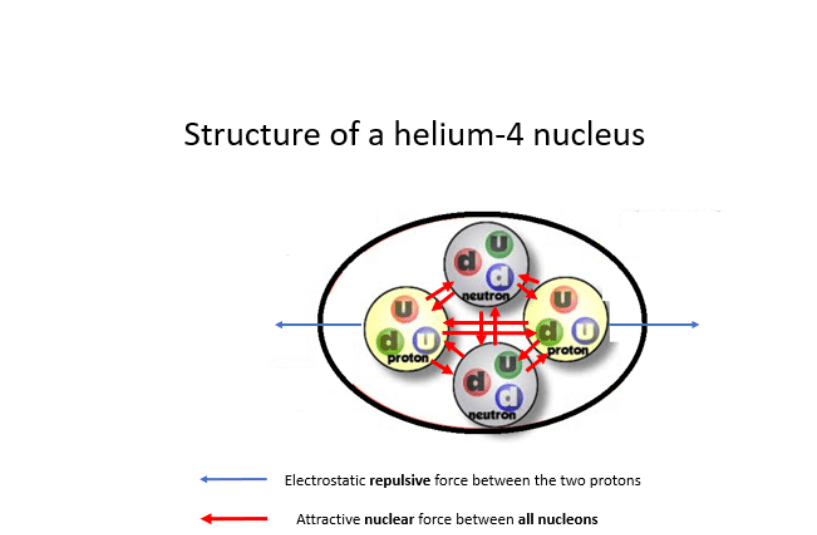
At the optimal distance of 10-15 m the attractive nuclear force between two neighbouring nucleons is 100 times stronger than the repulsive force between two neighbouring protons. The nuclear force enables nucleons to be attracted to each other forming atomic nuclei containing many protons and neutrons. However, its short range limits the size of atomic nuclei.
Consider a large nucleus, such as that shown below, then take a single proton A. This proton will ONLY be attracted to its neighbouring nucleons B, C, D, E, F and G by the nuclear force. The other nucleons in the nucleus will be too far away to have any significant attractive force. However, proton A will feel a repulsive electrostatic force from ALL protons in the nucleus.
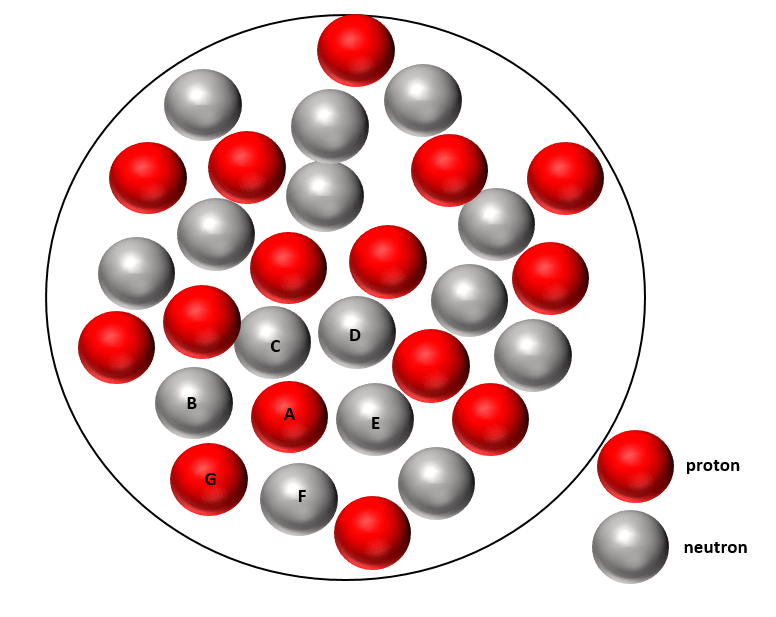
Section through a large atomic nucleus showing protons in red and neutrons in grey. (This is an oversimplification because protons and neutrons reside in shells within the atomic nucleus.)
As more protons are added to an atomic nucleus, the combined electrostatic force of all the existing protons repelling each other makes the nucleus more susceptible to radioactive decay. The heaviest stable nucleus is an isotope of lead, lead- 208, which has 82 protons and 126 neutrons. All atomic nuclei heavier than this decay radioactively. The heaviest atomic nucleus which is found naturally on Earth is an isotope of uranium which has 92 protons and 146 neutrons. It is radioactive but has a half-life of 4.5 billion years – roughly the same as the age of the Earth. All elements heavier than uranium are not found in nature and when we get up to nuclei with more than 100 protons, they are very unstable. The only known isotope of the final element in the periodic table organesson, which has 118 protons, has a half-life of less than one millisecond.

Elements shaded in white have at least one stable isotope. Elements shaded in dark yellow only have radioactive isotopes. The half-life of bismuth (Bi) 209 is so long (2 x1019 years) that it is shown as stable. The elements after Uranium(U) (bordered in red are all synthetic).
What would our Universe be like if the nuclear force were weaker?
Barrow and Tipler [1] calculated that in an alternative universe where the nuclear force were three times weaker than its current strength and the electrostatic force the same strength, rather than lead (element 82) being the heaviest stable element and an isotope of uranium being reasonably stable, (a half-life of 4.5 billion years counts as reasonably stable !) all elements heavier than atomic number five would be unstable. Key elements such as carbon, nitrogen and oxygen would not exist and the complex chemistry on which life is based would not be possible.

The first 15 elements
However, the nuclear force doesn’t even have to be three times weaker to prevent the development of life. Barrow and Tipler also point out that if the nuclear force were only 30% weaker than its current strength, then this would have two significant effects. Firstly, the nuclear fusion reactions in stars would generate significantly less energy. But even more importantly, deuterium also known as heavy hydrogen, which has a nucleus of one proton and one neutron would be unstable. Deuterium is an important component in the nuclear reactions which create odd numbered elements such as: nitrogen, phosphorus and sodium. Without deuterium, the abundances of these odd-numbered elements, which are essential for the chemistry of life, would be exceedingly small.
What would our Universe be like if the nuclear force were stronger?
If the nuclear force was 13% stronger, and the electrostatic force the same strength, the diproton which has a nucleus of two protons and no neutrons, (i.e., a helium-2 nucleus, would be stable)[1]. In this scenario, shortly after the big bang, virtually all the hydrogen in the early Universe, would have been converted to helium-2. Deprived of their hydrogen fuel there could be no long-lived stable stars and there would be no hydrogen compounds (such as water) which are essential for life. To put it simply if the nuclear force were only 13 % stronger, we wouldn’t exist.

Recent work has questioned whether such a small increase would be sufficient to convert all the hydrogen into helium-2 . A 2009 paper suggested that, rather than a mere 13%, a 50% increase in the relative strength of the nuclear force, would be needed to mop up nearly all the hydrogen into helium-2 in the early Universe [2] . Even so, it remains the case that a relatively modest increase in the relative strength of the nuclear force would mean that our Universe could not support life.
Amount of matter in the Universe
The average matter density in the Universe is approximately 3 x 10 -27 kg/m3 , of which roughly 85% is dark matter, the nature of which is unknown. Dark matter can be thought of as the scaffolding of the universe. The visible matter out of which all objects we can see (planets, stars, galaxies etc) collected inside this scaffolding and eventually formed stars and galaxies. Our current theories of galaxy formation indicate most that matter in Universe must be dark matter for this to have occurred.
However, if 99% (rather than 85%) of the matter of the Universe were in the form of dark matter, there wouldn’t be enough ordinary matter for stars and galaxies to have formed.
The Anthropic principle
The fact that our Universe appears to be finely tuned to allow for our existence is sometimes expressed as the anthropic principle. There are essentially two different versions of this, the strong anthropic principle and the weak anthropic principle and to complicate matters further there are differing definitions for each one. The definitions I’ll use in this post are from Barrow and Tipler.
The weak anthropic principle
The observed values of all physical and cosmological constants are not equally probable, but they take on values restricted by the requirement that carbon-based life can evolve, and that the Universe be old enough for it to have already done so.
One interesting fact about this is that it highlights that we observe the universe to be 13.8 billion years old. If the Universe were much younger, given all the complex steps which must have previously happened for Earth-like planets to exist around a Sun-like star and the additional 4.6 billion years for intelligent lifeforms to have emerged on this planet there wouldn’t been enough time for this to have occurred.
Generally speaking, the weak anthropic principle is accepted by most astronomers. In fact, one criticism of it is that it isn’t a scientific theory. Rather, it is a tautology – a statement which must be true.
More controversial is a strong anthropic principle.
The strong anthropic principle
The Universe must have those properties which allow life to develop within it at some stage in its history.
Or to put it another way, without life you don’t have a Universe!
A variation on this is the participatory anthropic principle stated by John Archibald Wheeler, that in some sense you can’t have a universe without observers to observe it. This was based on a particular interpretation of quantum mechanics
The participatory anthropic principle
Observers are necessary to bring the universe into being
Related Posts
I hope you have enjoyed this post. It is part of series of three (listed below) about cosmic fine tuning.
- The fundamental forces and fundamental constants which govern the Universe have been in some sense finely tuned to allow for our existence.
- Examples of cosmological fine tuning. How different our Universe would be if these four forces had very different relative strengths.
- Why is our Universe finely tuned?
A video of this post is also available
References
[1] Barrow, J.D. and Tipler, F.J. (1986). The anthropic cosmological principle. Oxford Oxfordshire ; New York: Oxford University Press p322-324
[2] MacDonald, J. and Mullan, D.J. (2009). Big bang nucleosynthesis: The strong nuclear force meets the weak anthropic principle. Physical Review D, 80(4). doi:10.1103/physrevd.80.043507.
Discover more from Explaining Science
Subscribe to get the latest posts sent to your email.

[…] discussed in my previous two posts our Universe appears to be very finely tuned and is governed by four fundamental […]
LikeLike
Just an additional thought on finely-tuning:
My Dinosauric Principle, which to me is the same as the Anthropic Principle, could be an argument for a finally-tuned universe, but happily enough, I think, capriciousness cannot be excluded.
LikeLike
My watch is finely-tuned to the most perfect state possible, showing the exact time twice a day. It has stopped.
By the way, in the “Composition of our Universe” I miss ordinary energy.
LikeLike
🙂
LikeLike
Much enjoyed this Steve! Worrying about nucleon G in that diagram being red ie. a proton, so wouldn’t proton A be repelled by it and vice versa? Just can’t quite get head around that bit but will try again. Nice cliffhanger ending!
LikeLiked by 1 person
Hi Margaret thanks for your comment….
And yes proton G is repelled from proton A, due to the electrostatic force. However, the key thing here is that because A and G are so close to each other the nuclear force binding them together is 100 times stronger. So that will easily overcome the electrostatic repulsion between A and G.
As I said in the article the nuclear force is so short range it only acts on nucleons which are effectively adjacent. In comparison, the electrostatic force has a much longer range.
Hope this helps…
Steve
LikeLike
Reply to Erhan (“Are we absolutely sure there are only four forces…?)
No, of course we aren’t. Tomorrow there could be a completely unexpected discovery explainable only by invoking a fifth (or more) force. A few years ago a fifth fundamental force (tentatively named Quintessence) was proposed as a possible basis for Dark Energy. However at present, four forces appear to be enough to develop theories at the frontiers of particle physics & cosmology (such as Quantum Gravity; a workable theory of which is still not quite with us). But it’s possible that if & when we investigate the properities of some exotic states of matter – such as at the center of a neutron star – we may have to accept (reluctantly) the need for further fundamental forces.
LikeLiked by 1 person
Hi Steve,
As usual, thanks for all the hard work (not forgetting the contribution of Barrow & Tipler of course!).
Two comments (not criticisms, I hasten to add):
1) In the table “The first 15 elements”, we have several that are merely ‘trace’ or ‘important’. i.e. Lithium, Boron, Fluorine, Silicon. We could put several others in these two minor categories of course: zinc, selenium etc. And notably, iodine is vital for animal life – in this particular instance; on this planet.
But we’re discussing the Universe generally. Given enough time, evolution is very persistent & very ingenious and we do it a disservice if we assume that it wouldn’t have managed without all of these ‘marginals’ if they’d been unavailable for some reason.
2) The “participatory anthropic principle”. This is too much to put up with! Do its advocates seriously expect us to believe that the Universe didn’t exist in the eras of the CMB, of recombination, of initial star formation etc.? – which there’s more than adequate evidence of. And further forward in time, what type of creature constitutes an observer? A human no doubt, but what about a more modest level of consciousness: another primate? – an insect? Maybe they allowed the Universe to come into being gradually, waiting for humans – and their superiors – to finish the job!
I suppose there’s an alternative that’s even more unsavoury: that the presence of human observers somehow exerted an influence backwards in time & allowed the Big Bang to happen??
Regards, David.
LikeLike
Thank you David for some interesting observation and yes it is fair to say that only a small fraction of cosmologists believe in the Participatory Anthropic Principle!
Steve
LikeLike
Are we absolutely sure that there are only 4 forces…?
LikeLike
What a super article – and the one before it. Brilliant.
Cheers
Barbara
>
LikeLike
Fantastic and fascinating article!
LikeLike
Thank you
LikeLike
[…] Source link […]
LikeLike