Updated 26 November 2025
This is first of a series of three posts in which I’ll talk about the idea that the fundamental forces which govern our Universe and fundamental constants such as: the strength of gravity, the electric charge of the electron and the amounts of matter, dark matter and dark energy in the Universe have been in some sense finely tuned to allow for our existence.
This idea was discussed by Stephen Hawking in his 1988 book: A brief history of time [1].
The laws of science, as we know them at present, contain many fundamental numbers, like the size of the electric charge of the electron and the ratio of the masses of the proton and the electron.
… The remarkable fact is that the values of these numbers seem to have been very finely adjusted to make possible the development of life.’
It has been covered in many other works in recent years. One of the most popular is Just Six Numbers [2] by Martin Rees who was UK’s Astronomer Royal from 1995 until 2025.
The book makes the interesting point that, if any of six key numbers were very different, then the Universe couldn’t exist in its current form. I have listed Martin Rees’s six numbers on a reference page on my blog

What we know about the Universe
Before we discuss fine tuning further it’s worth summarising what we know about the Universe. The general consensus is that it’s 13.8 billion years old and began in a state of exceedingly high density (the Big Bang) and has been expanding ever since. Averaged-out the Universe is made up as follows.
- 5% ordinary matter. All the objects we can directly observe (e.g. stars, planets, galaxies and ourselves!) are made from ordinary matter.
- 27% dark matter. This is a form of matter which has never been observed directly, but there are strong reasons for believing it must exist.
- 68% dark energy . It is dark energy which causes the expansion of the Universe to speed up.

The universe is governed by four fundamental interactions, which are: electromagnetism, gravity, the strong interaction, and the weak interaction.
Electromagnetism
The electromagnetic force acts on electrically charged particles. Electric charge can be either positive or negative. If two particles both have a positive charge or both have a negative charge, there is a repulsive force. If one is negative and the other positive there is an attractive force. Particles having zero charge experience no force.

The magnitude of the force F between two electric charges Q1 and Q2 separated by a distance R from each other is given by the expression below.
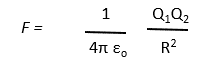
If the charges are measured in coulombs, the standard unit of electric charge, and the distance in metres, then the force will be in newtons. (One newton is roughly the force that a 100 gramme object would experience due to the Earth’s gravity.)
Those of you who have studied physics at high school may recall this relationship is called Coulomb’s law. The term (1/4πεo) is called the Coulomb constant and has the value 8.988 x 109 in standard units. This force between two charges is usually called the electrostatic force and occasionally the Coulomb force.
Coulomb’s law shows that the force between two charges falls off as the square of the distance between them: a relationship known as the inverse square law. So, if the distance between two charges doubles, the force is reduced by a factor of four.
It is this electrostatic force which holds atoms together. It binds the negatively charged electrons into orbits around the positively-charged atomic nucleus.
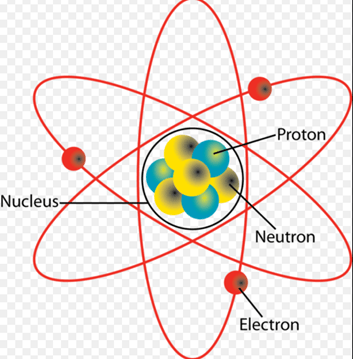
The diagram above shows a representation of a lithium atom. Three negatively charged electrons are in orbit around the nucleus, which consists of three positively charged protons and four neutrons which have zero electric charge. In reality, although diagrams like this are widely used, they are oversimplifications. Electrons do not orbit the nucleus in circular orbits. Instead, they are found in orbitals defined by the rules of quantum theory, which is far too large a topic to cover in this post!
The electrostatic force is responsible for chemical bonding, which causes atoms to combine into chemical compounds.

The forces between molecules, which are much weaker than chemical bonds between the atoms within molecules, are also a result of the electrostatic force. The structure of liquid water is shown below.
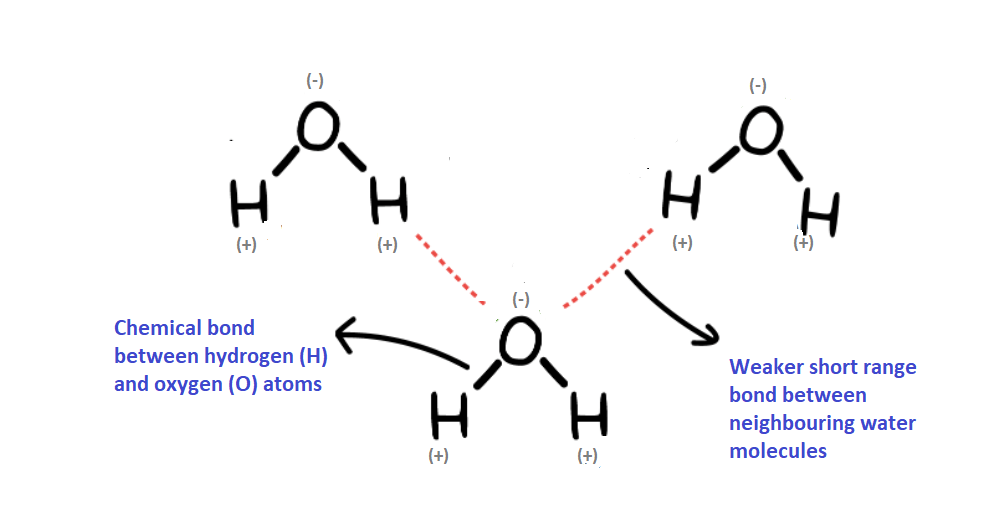
As a whole a water molecule has no net electric charge. However, within the water molecule, the oxygen atom has a small negative charge and the two hydrogen atoms each have a small positive charge. This causes a weak force between the oxygen atom of a water molecule and the hydrogen atoms of its neighbours
This weak intermolecular force (which is called hydrogen bonding) means that water is a liquid at room temperature. Energy needs to supplied to break these hydrogen bonds and convert it into steam. In glass, silicon and oxygen atoms are held together by stronger chemical bonds.

The forces between water molecules are one example of intermolecular forces. Essentially everything we see in our daily lives on a small/medium scale is held together by electrostatic forces. This includes the molecules in the cells which make up our body!
Electric currents, magnetism and electromagnetic radiation.
An electric current occurs when electrically charged particles (such as the electrons in a wire) move.
- When an electric current flows, it induces a magnetic field.
- When a magnetic field changes it induces a voltage in a loop of wire
It was one of the greatest achievements in nineteenth century science, for James Clerk Maxwell and Michael Faraday to unify electricity and magnetism, which had been thought of as being separate phenomena, into electromagnetism. Maxwell was able to show that electromagnetic radiation (such as light) was due to changing electric and magnetic fields.
Gravity
Gravity acts on anything with mass and is always attractive. Like the electrostatic force it follows an inverse square law. The magnitude of the force (F) between two masses M1 and M2 separated by a distance R from each other is given by:

If the masses are measured in kilograms and the distance in metres, then the force will be in newtons. G is known as “the gravitational constant”, and is often called “Big G”. It has the value 6.674 x 10-11 in standard units.
Gravity is extremely weak compared to the electrostatic force. If we take the example of a hydrogen atom which consists of a single proton orbited by a negatively charged electron, the mass of the proton is a miniscule 1.672 x 10-27 kilograms and the mass of the electron even smaller at only 9.109 x 10-31 kilograms. If we assume that the proton and electron are 10-10 metres apart, which is their typical separation in a hydrogen atom, then the gravitational force between the electron and proton is an unimaginably small 10-47 newtons.

If we now consider the electrostatic force between the proton and electron, the proton has a positive charge of roughly 1.6 x 10-19 coulombs and the electron has a negative charge of the same magnitude. The electrostatic force between the proton and electron is 2.3 x 10-8 newtons which is 39 orders of magnitude greater than the gravitational force.
This illustrates that on atomic scales gravity is so weak it can be totally ignored. Even on the scales we are used to in our everyday life gravity is insignificant. For example, if we take two one kg masses and place them 1 metre apart, the gravitational force between them is only 0.000 000 000 067 newtons. It is only on much larger scales that the effects of gravity become significant.
If we go to much larger objects and consider the Earth and the Moon. The Earth has a mass of about 5 972 000 000 000 000 000 000 tonnes (5.972 x 1024 kg), the Moon has a mass roughly eighty times smaller and the mean distance between the Earth and the Moon is roughly 384 000 km. The gravitational force between the Earth and the Moon is roughly 2 x1020 newtons. It is this gravitational force which keeps the Moon in orbit around the Earth.

Because the amount of positive and negative charge cancel each other out, the Earth and the Moon have almost zero net electric charge and the electrostatic force (which only acts on charged bodies) is zero. As I’ll explain next the nuclear force and the weak interactions are very short range and it is only gravity which matters at large distances.
The Strong Interaction (a brief overview)
The strong interaction only acts on particles which have a property which particle physicists have labelled colour charge. This has nothing to do with the everyday meaning of colour it is just a label for this property.
Colour charge can have six possible values, the three ordinary colours red, blue and green and the three anti-colours anti-red (normally represented as cyan), anti-blue (normally represented as yellow), and anti-green (normally represented as magenta). It is described in detail by a mathematically very complicated theory called quantum chromodynamics, but in essence there is an attractive force between particles of a different colour charge.
In quantum chromodynamics, the strong interaction is carried by particles called gluons, which themselves have colour charge.
Protons and neutrons found in atomic nuclei are not fundamental particles. They are composed of quarks. There are six type of quarks (known as flavours) which physicists have given the rather odd names of: up, down, charm, strange, top and bottom. Only the up and down flavours are stable. Quarks of the other flavour rapidly decay into an up or a down quark. All quarks have a colour charge which can be red, blue or green.
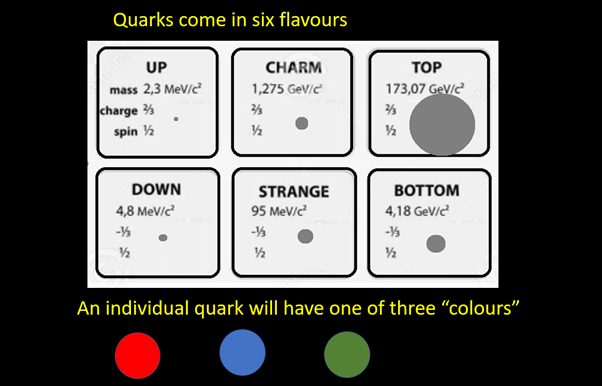
There are also six flavours of antiquark: anti-up, anti-down, anti-charm, anti-strange, anti-top and anti-bottom. Each antiquark will have a colour charge which can be anti-red, anti-blue or anti-green.

Isolated quarks are never found. Quarks are always tightly bound together by the strong interaction into particles called hadrons which contain two or more quarks. Protons and neutrons (which collectively are known as nucleons) are hadrons which contain three quarks
- Protons are made up of two up quarks and one down quark. These quarks are constantly changing colour but, at a given instant in time, there’s always one red, one blue and one green quark within a proton. Red, blue and green colour charges combine to make a colour charge of white, so the proton is colourless overall.
- Neutrons are made up of one up quark and two down quarks. Like the proton, one is red, one is blue and the other one is green. So, the neutron is also colourless overall.

The nuclear force
Because nucleons (i.e. protons and neutrons) have no overall colour charge, we might expect that there would be zero strong interaction between two nucleons. This is in general true. However, if two nucleons are very close together (around 10-15 metres) we do get an interaction between them. This occurs when the quarks of one nucleon interact with quarks having a different from a neighbouring nucleon (e.g. the red quark of proton will be attracted to the blue and green quarks of an adjacent neutron ). This is called the residual strong interaction or the nuclear force and is extremely short range, Its strength is effectively zero at a distance of 3 x 10-15 metes. It is the nuclear force which binds protons and neutrons together into atomic nuclei.
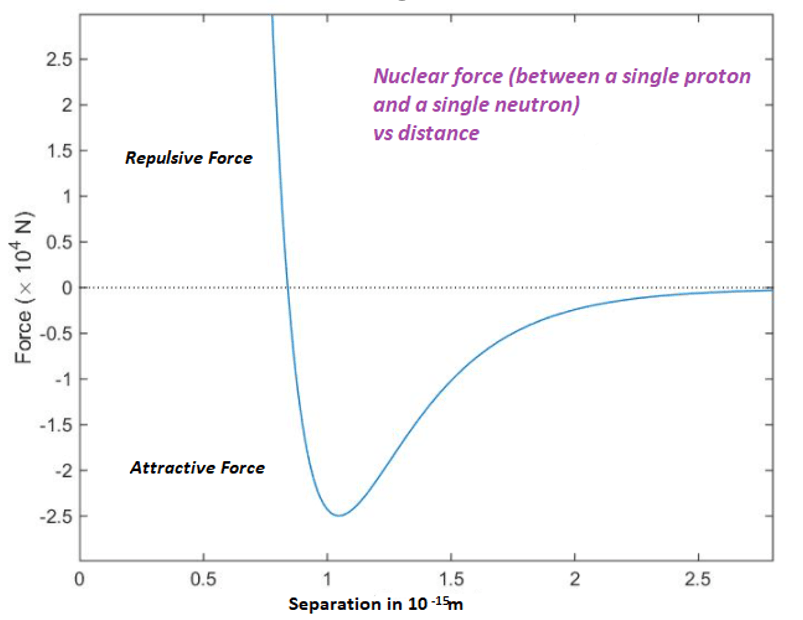
The short-range nuclear force between two nucleons which have zero colour charge is analogous to the intermolecular force between adjacent molecule which have zero net electric charge.
Weak interaction
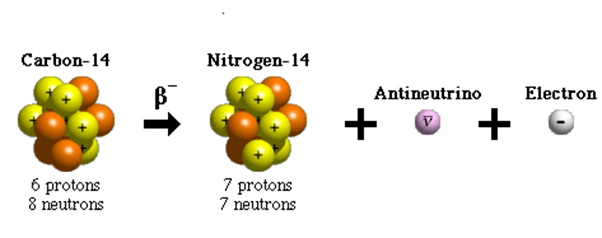
The weak interaction only acts over very short distances – around 10-17 metres. It is the only interaction which can cause a quark to change flavour. So, for example, an up quark can become a down quark or vice versa.

One of the most notable examples of beta decay is when a carbon 14 nucleus changes into a nitrogen 14 nucleus. The half-life of this decay is 5700 years and it is often used for radiocarbon dating
Related Posts
I hope you have enjoyed this post. It is part of series of three (listed below) about cosmic fine tuning.
The fundamental forces and fundamental constants which govern the Universe have been in some sense finely tuned to allow for our existence.
Examples of cosmological fine tuning. How different our Universe would be if these four forces had very different relative strengths.
Why is our Universe finely tuned?
This post is now available as a video
References
[1] Hawking, S. (1988). A Brief History of Time: From the Big Bang to Black Holes. London: Bantam Books.
[2] Rees, M.J. (2015). Just six numbers : the deep forces that shape the universe. London: Weidenfeld & Nicolson.
Discover more from Explaining Science
Subscribe to get the latest posts sent to your email.

[…] discussed in a previous post the chemical bonds binding atoms together into molecules, on which the whole of the science of […]
LikeLike
[…] Source link […]
LikeLike
Thanks Steve: a superb explanation.
Regarding the weak-interaction Feynman diagram, it might be worth mentioning why the arrow-head for the anti-neutrino points “inwards”. i.e. it could look as though the anti-nu is being absorbed rather than emitted – which of course it isn’t.
Regards, David.
LikeLiked by 1 person
Thank you david. You make a good point about the Feynman diagram
LikeLike